Medicine China claims it uses as cure for COVID-19 reaches Manila
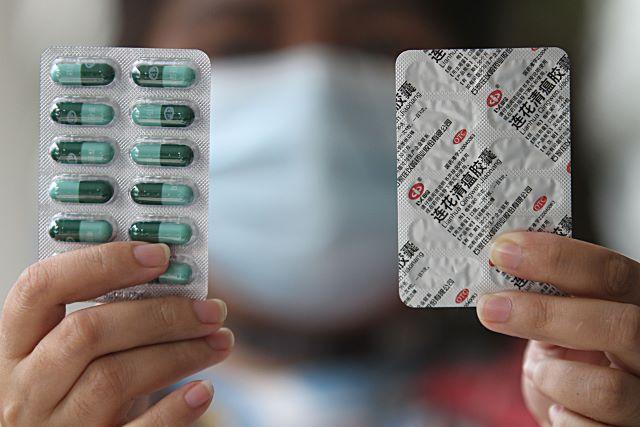
A woman in Manila on Thursday, August 13, 2020 shows stock of traditional medicine supposedly used in China as COVID-19 treatment called "Lianhua Qingwen."
However, the Philippine Food and Drug Administration (FDA) has only given Lianhua Qingwen -- a prescription drug -- a certificate of product registration, but not yet as cure for coronavirus disease.
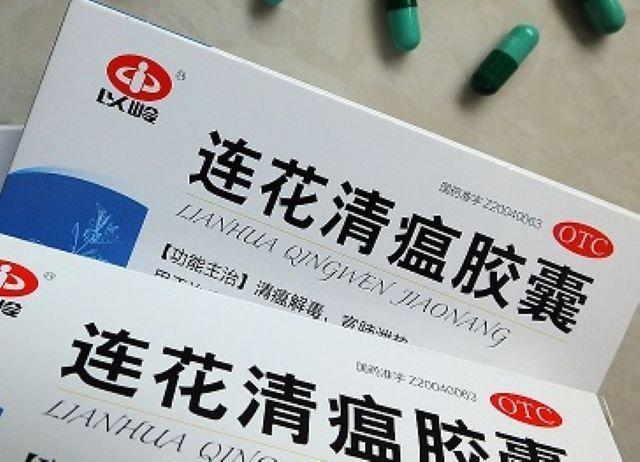
On Wednesday, the FDA announced that it has approved "China's COVID-19 drug" for Philippine use to treat lung toxins, fever, and other related symptoms.
FDA Director General Eric Domingo, however, said that the Lian Hua QingWen has yet to be approved in Philippines as a treatment for the coronavirus disease.
The drug’s Certificate of Product Registration for the Philippines indicated that it could be used to combat heat-toxin invasion of the lungs, fever, aversion to cold, muscle soreness, and stuffy and runny nose.
“It was approved last Friday as traditional medicine. It may now be sold, prescribed, and used in the Philippines,” Domingo told GMA News Online.
A report from China Global Television Network (CGTN) on Wednesday said that the Chinese Embassy in the Philippines welcomed the approval of the popular traditional Chinese medicine (TCM) Lianhua Qingwen, a treatment for the novel coronavirus disease, marking an important progress in the entry of TCM products into the Philippine market.
According to the Embassy, the Lianhua Qingwen capsule is an approved COVID-19 treatment for mild and moderate patients in China, as well as in China's Hong Kong Special Administrative Region (SAR) and China's Macao SAR, Brazil, Indonesia, Canada, Mozambique, Romania, Thailand, Ecuador, Singapore and Laos, the CGTN report said. —with a report from Danny Pata/LBG, GMA News




